ISO 13485 Certified: IEC 62304 Compliant
Medical Device Software & SaMD Development — Built Faster
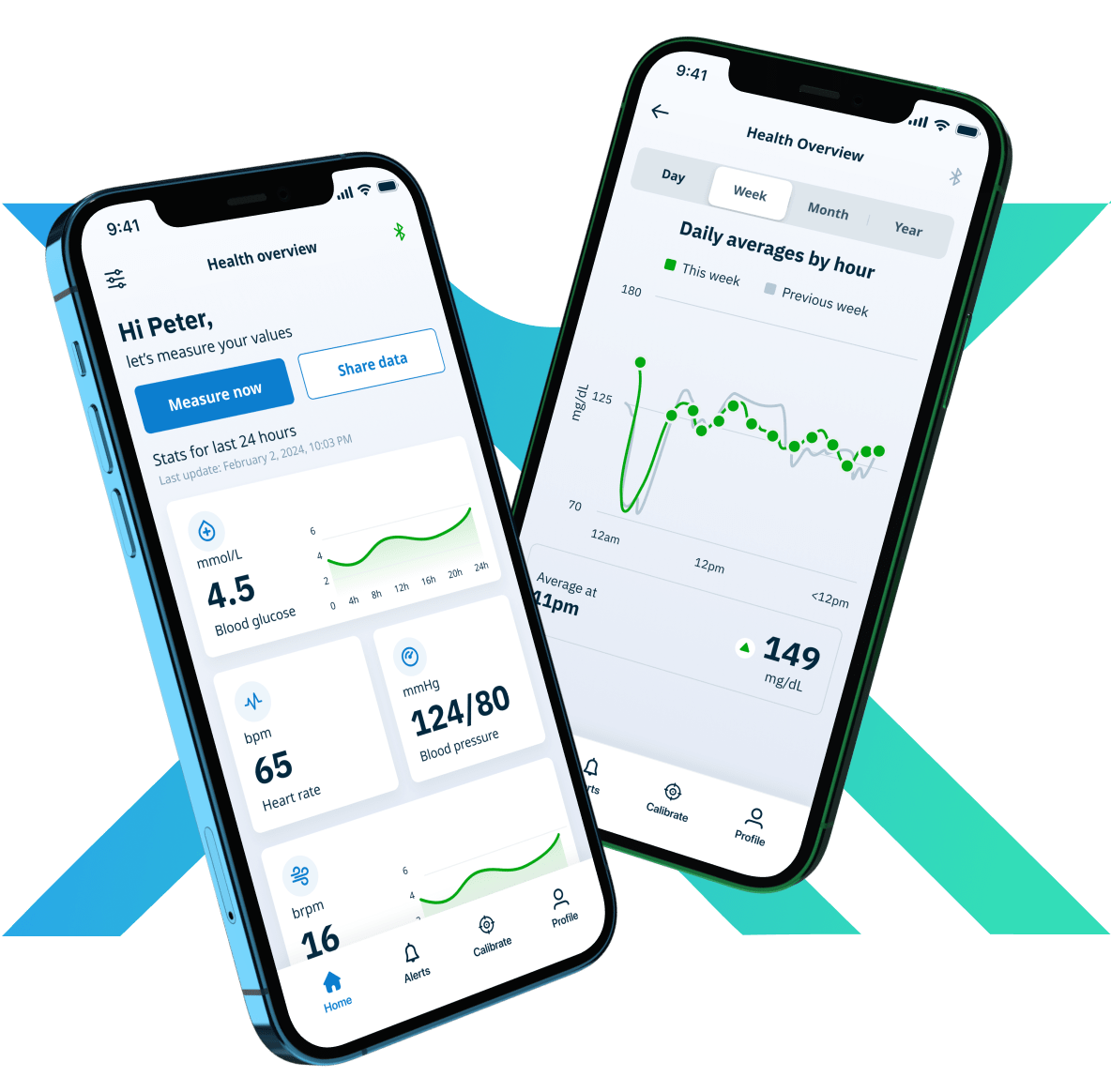
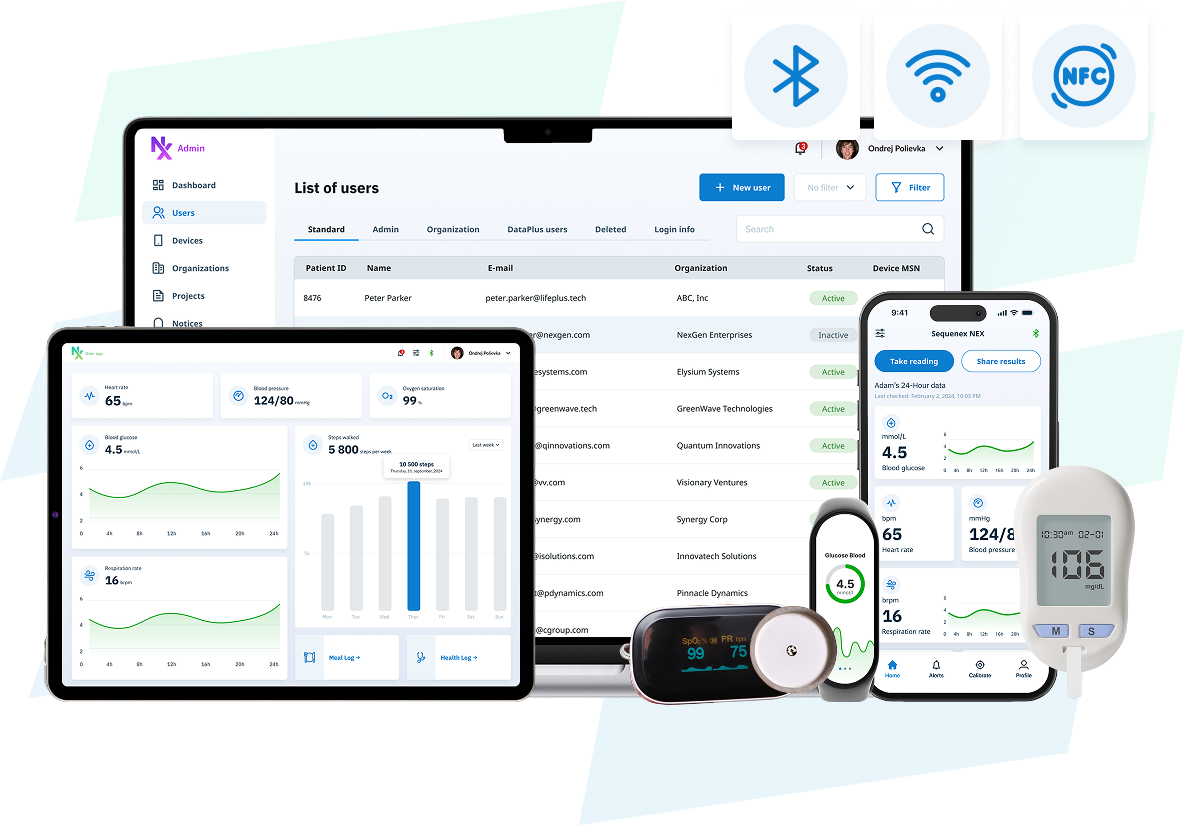
NEX Platform — A Prebuilt Software Foundation for Medical Devices and SaMD
The NEX Platform delivers six to nine months of engineering scaffolding prebuilt — iOS and Android apps, patient and clinician dashboards, an administrator portal, and secure cloud infrastructure for medical data — engineered to function as a unified system, with the IEC 62304 and ISO 13485 engineering scaffolding already in place.
Reduce product development time, complexity, and costs with a prebuilt and customizable software foundation for regulated medical devices and SaMD. Your team inherits the engineering scaffolding instead of recreating it.
Built to function as a unified system, NEX includes iOS and Android apps, Patient and Clinician Dashboards, an Administrator Portal, and Secure Cloud Infrastructure for medical data.
Our Services
Biosensor IntegrationsSequenex is a full-service software engineering and product development firm for connected medical devices, biosensors, CGM, and SaMD.

Medical Device Software Testing, QA and V&V
Medical device software verification and validation done right the first time — automated and manual testing across mobile, cloud, and connected devices, with full traceability to requirements that holds up under audit.

AI and ML for Medical Device Software
AI and ML for medical device software, engineered for the regulated path — predictive analytics and algorithm development designed with traceability, V&V, and FDA AI/ML guidance familiarity built into the engineering approach. Engineering deliverables structured to integrate with your regulatory team’s submission package.

Software Product Development
Connected medical device software development — mobile, cloud, and real-time data — built end-to-end under the rigor of our ISO 13485-certified QMS to IEC 62304, so the engineering deliverables your regulatory team needs feed into your DHF and submission package as you build, not after.

BLE, NFC, and WiFi for Connected Medical Devices
BLE, NFC, and WiFi connectivity for medical devices — pairing, data streaming, and protocol handling that just work, integrated cleanly into your iOS and Android apps and your cloud backend.End-to-end implementation of device-to-app communication, including pairing, data streaming, and protocol handling for connected medical devices.

MedTech Resourcing
MedTech software engineers and dedicated software development teams, ready to plug in. From a single engineer filling a skills gap to a fully managed team for SaMD or connected device development — trained in a regulated software discipline through Sequenex’s SaMD-Trained Engineer program.

AI-Assisted Engineering
AI-assisted medical device software development that reaches submission faster without losing the audit trail — engineer-controlled use of AI for development, documentation, and testing, under the rigor of our ISO 13485-certified QMS.

Medical Device Cybersecurity & Data Protection
Medical device cybersecurity built in from architecture, not bolted on at submission — encryption, RBAC, and audit trails across device, mobile, and cloud, aligned to FDA premarket cybersecurity guidance, HIPAA, and GDPR.End-to-end security across device, mobile, and cloud, including encryption, RBAC, and audit trails, supporting HIPAA and GDPR-requirements.

Custom Firmware for Medical Devices
Custom firmware for biosensors and connected medical devices that holds up in the real world — optimized performance, the features your roadmap actually needs, and connectivity that doesn’t drop in the field.
Sequenex’s software development process is IEC 62304-compliant from the first sprint, with ISO 14971 risk management running in parallel — so when audit day comes, the documentation is already there.
- IEC 62304 software lifecycle, fully documented
- ISO 14971 risk management at every stage
- Regulatory requirements traced to source
- Full traceability and accountability across the SDLC
A Design History File (DHF) is the regulatory record of a medical device’s design and development, required by FDA 21 CFR 820.30 and ISO 13485. Most teams scramble to assemble theirs at submission. Sequenex delivers the complete software documentation package — software development plans, requirements, design specs, V&V records, and risk management files — already structured to integrate with your quality team’s DHF, aligned to design controls. So submission week is paperwork, not a fire drill.
ISO 13485 Certified QMS
Our ISO 13485-certified QMS underscores our commitment to engineering discipline in medtech software, ensuring your projects are delivered with the documentation traceability and audit-readiness regulated software requires.
Our Expertise
Sequenex develops software products for connected medical devices, biosensors, and SaMD—covering architecture, integration, testing, and risk management.

SaMD Compliance and Development Processes
SaMD development under ISO 13485 and IEC 62304 is what we do every day — not a sideline. Every project is engineered under the rigor of our ISO 13485-certified QMS, with the documentation discipline and regulatory alignment your customer-facing submission depends on built into the workflow rather than reviewed at the end. Specialization in SaMD, with engineering aligned to ISO 13485 and IEC 62304 standards.
Testing and Quality Assurance
Medical device software testing that holds up under audit — unit, integration, and acceptance testing across system and subsystem level, run through automated pipelines with traceable test case management mapped to your requirements.Expert in rigorous system and subsystem testing — including unit, integration, and acceptance testing. Emphasizes automated testing, comprehensive test case management, and regulatory standards for quality solutions.
Companion Apps Development
Companion apps for connected medical devices — remote patient monitoring (RPM), medication adherence, chronic disease management, wearables, and data analytics — built as iOS and Android applications integrated with biosensors, CGM systems, and clinician dashboards.Remote Patient Monitoring, Patient Monitoring, Fitness & Wellness, Medication Adherence, Chronic Disease Management, Wearables, Data Analytics & Reporting
Risk Management
Medical device software risk management under ISO 14971, applied from concept through post-market surveillance — hazard analysis, risk assessment, and mitigation engineered to feed cleanly into your regulatory team’s risk management file.
Diabetes and Biosensor Subject Matter Experts
Diabetes and biosensor software is where our team holds its deepest subject matter expertise — CGM, insulin pump, and connected pen integration are the categories we know best. You get robust, integration-ready software from a team that already understands where the sharp edges live across these product types.
Engineering Aligned to Regulatory Frameworks
At Sequenex, the engineering discipline that supports your regulatory team’s work — IEC 62304 software lifecycle deliverables, ISO 14971 risk management, and DHF-ready documentation — is built into every step of development. Your transition from concept toward commercial release moves on a steady cadence because the engineering deliverables your regulatory and quality team needs are already in their formats, ready to integrate with their submission package.
- Test-driven development
- Automates testing procedures
- Simulates device behavior
- Ensures reliability and performance
- Mobile App Persistence Library: Ensures data integrity
- Graphing Library: Visualizes medical data
- Logging Library (HIPAA-ready): Secure logging architected for HIPAA controls
- OIDC/OAuth2 Authentication: Robust authentication mechanisms
- Safeguards sensitive patient information
- Automated Documentation: Generates comprehensive engineering documentation that integrates with your regulatory team’s submission package
Tools and Solutions
We use advanced solutions, tools, and processes to accelerate medtech and digital health software delivery. We support rapid development and regulatory alignment for a faster path to submission.
Knowledge Hub
SaMD News & Industry Updates.
Explore Knowledge Hub
May 1, 2026
Apple App Store Medical Device Compliance: How This New Rule Changes the Digital Health Go-to-Market Strategy
Apple’s new medical device status requirement signals a shift in digital health compliance, where app stores actively influence regulatory readiness.…
April 30, 2026
Building CGM Software in 2026: Market Outlook, Software Trends, and Regulatory Realities for Hardware Companies
Discover key trends, growth drivers, and strategic insights shaping the global CGM market in 2026. From non-invasive tech to digital…
April 20, 2026
Wellness vs Medical Device Regulation: The Line Is Thinner Than You Think
Wellness apps and medical devices are built on the same physiological signals—the difference lies in intent and claims. As the…